
Inside the Phonak Audiology Research Center, USA
A PARC series highlighting Phonak research centers worldwide, where clinical questions become clinical evidence.
The Phonak Audiology Research Center (PARC) opened in 2014 in the heart of the largest market for Phonak technology. Organizationally part of Sonova Global R&D and located at the company’s US headquarters near Chicago, PARC works closely with engineering teams in Switzerland and collaborates with local experts in clinical training, marketing, and audiology education.
These connections allow PARC to respond quickly to emerging clinical questions and customer needs. The result is research that directly informs product design, clinical guidance, and ultimately the experience of hearing aid users.
Led by VP of Clinical Research Strategy Jason Galster, the multidisciplinary PARC team brings together expertise in audiology, speech and hearing science, linguistics, engineering, psychology, and data science, ensuring research questions are examined rigorously and from multiple scientific perspectives.

| Center Snapshot Location: Aurora, Illinois, USA Founded: 2014 Team size and disciplines: Audiologists, research scientists, data scientists, clinical operations and compliance specialists Core Capabilities: ⦁ Controlled speech-in-noise testing ⦁ Multi-speaker acoustic scene simulation ⦁ Clinical fitting and verification ⦁ Pre-market validation and post-market outcome studies ⦁ Real-world field evaluations |
Step inside PARC
Each research space at PARC is designed to answer a different question, from how devices perform under controlled conditions to how people experience sound in everyday life. Together, these environments ensure that innovation is both scientifically rigorous and clinically meaningful.
Inside the lab
Sound-treated booths
In this space, precision is everything. Three advanced sound-treated booths allow precise measurement of speech understanding and listening effort under tightly controlled acoustic conditions.

These booths include:
- Clinical audiometers (Otometrics Madsen)
- 12 loudspeaker arrays
- Professional sound-mixing software for complex acoustic scene creation
These setups enable simulation of challenging, multi-directional listening environments while maintaining strict experimental control. This level of precision strengthens the reliability of findings before innovations move toward market readiness.
Programming and consultation rooms
Research only matters if it translates into real-world care. Two dedicated consultation rooms are designed to mirror everyday clinical environments, ensuring that findings are directly applicable to how hearing care professionals work. They include:
- Audioscan Verifit2 and Otometrics Aurical verification systems
- Noahlink Wireless programming

By integrating research protocols into familiar clinical workflows, PARC ensures that study outcomes are directly applicable to everyday practice.
Dedicated Lyric room
Some innovations require specialized environments. This space enables detailed evaluation of extended-wear solutions within a controlled and comprehensive research framework. The dedicated Lyric room allows:
- Lyric sizing and fitting
- Cerumen management
- Ear mold impressions
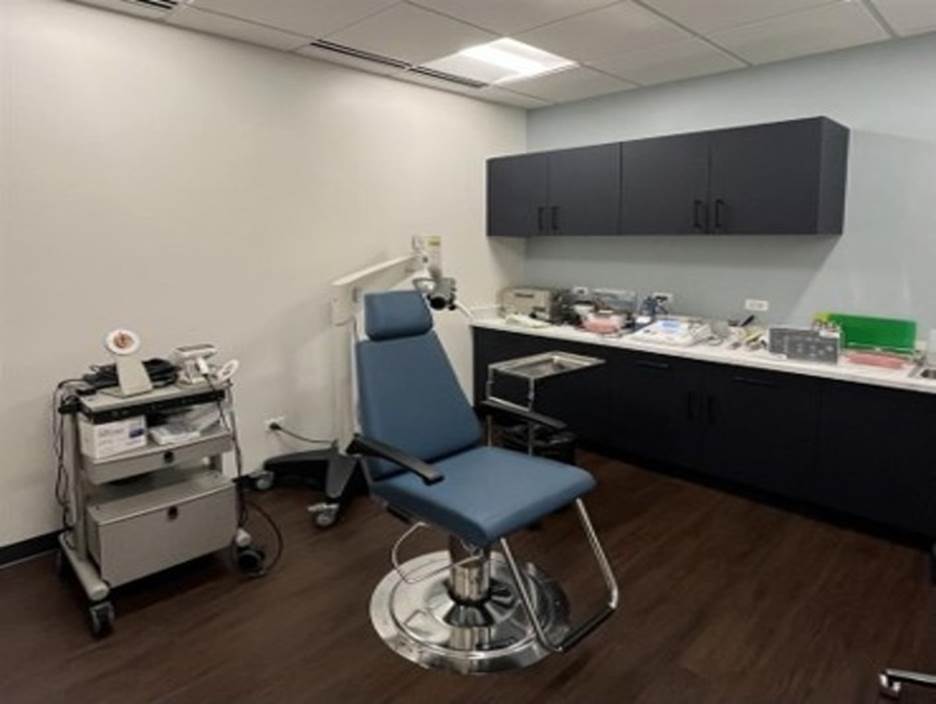
This capability supports evaluation of extended-wear solutions within a comprehensive research framework.
The Listening Loft
Understanding real-world listening goes beyond the clinic. The Listening Loft is designed to recreate everyday environments, helping researchers evaluate how hearing solutions perform in conditions that feel natural to users.

- Apartment-style layout for ecological realism
- Controlled acoustic manipulation
- Measurement systems integrated into everyday scenarios
It offers greater ecological validity than a traditional booth, while still allowing acoustic manipulation and measurement control. This balance between realism and control enhances external validity and allows researchers to explore performance in conditions that closely reflect daily life.

Phonak Audiology Research Centers (PARC)
PARC is our global research center network dedicated to setting the highest standards in hearing aid performance testing, ensuring every innovation delivers real-world benefits for clients. With locations around the world— in the United States of America, China, Canada, and Switzerland – PARC’s global presence ensures that the diverse needs of hearing aid wearers are reflected and addressed.
Learn more phonak.com/evidence
Evidence of Excellence
PARC conducts investigations along the entire product development pathway, including:
- Early proof-of-concept work involving new innovations
- Pre-market studies to support product claims and establish clinical readiness
- Post-market clinical research involving state-of-the-art methodologies and a holistic perspective on hearing care focused on auditory wellness and overall health
In 2025 alone, PARC completed over 250 hearing aid fittings and drew from a database of more than 200 research volunteers, helping ensure studies reflect real clinical populations.
Beyond internal research, PARC collaborates with numerous universities and clinical partners across the United States, supporting both pediatric and adult audiology research. Findings are regularly presented at scientific conferences and published in peer-reviewed journals, contributing to the broader evidence base in hearing care. All research activities are conducted under strict compliance and ethical standards, reinforcing transparency and clinical credibility.
From lab to clinic
PARC research supports hearing care professionals by:
- Providing confidence that innovations have been rigorously evaluated in clinical populations
- Supplying evidence that clarifies expected benefits for clients
- Expanding understanding of hearing care beyond audibility toward listening effort, cognition, and overall well-being
Example: Hearing aid use and cognitive outcomes
In a recent exploratory investigation, PARC examined cognitive performance and interpersonal relationship outcomes in new hearing aid users.1
After six months of consistent device use, participants demonstrated:
- Improved performance on speech processing tasks
- Improved short-term memory scores
- Reduced self-reported hearing disability
- Reduced third-party disability reported by significant others
These findings support counseling conversations that extend beyond improved audibility by giving evidence-based language for the question clients often ask: “Will this make a difference in my day-to-day life?”
What is next
PARC continues to expand research in:
- Ecologically valid outcome measures
- Listening effort and cognitive load
- Holistic measures of auditory wellness
- Cross-disciplinary collaborations
As hearing care evolves toward outcome-centered practice, PARC remains focused on ensuring that innovation is consistently grounded in clinical evidence.
Together, Phonak research centers worldwide form a global network advancing evidence-based hearing care. Each center contributes local expertise and specialized methods to help translate research into clinical confidence.
To explore the full body of evidence behind PARC research, visit the Phonak Evidence Library and search by topic, feature, or product – Link
Reference:
- Adler, M., Stewart, E., Singh, G., Keller, M., & Seitz-Paquette, K. (2025, March 26-29). The impact of amplification on self-perceived handicap, third party disability and social participation [Poster presentation]. American Academy of Audiology Annual Conference, New Orleans, LA, United States.
